Preparing for laboratory compliance audits becomes overwhelming when documentation is scattered and training records are incomplete. Non-compliance leads to regulatory penalties, delayed research projects, and compromised data integrity. This guide provides a structured, step-by-step approach to implement and maintain laboratory standards confidently, helping biotech and pharmaceutical lab managers navigate ISO/IEC 17025 and Good Laboratory Practice requirements effectively.
Table of Contents
Key takeaways
| Point | Details |
| Understanding standards | ISO/IEC 17025 and GLP form the foundation for biotech lab compliance and regulatory acceptance. |
| Preparation essentials | Staff training, complete documentation, and equipment calibration protocols must be established before audits. |
| Quality control implementation | Validated analytical methods like HPLC and mass spectrometry verify peptide synthesis and ensure data integrity. |
| Documentation practices | Accurate Certificates of Analysis and batch records support audit readiness and traceability. |
| Troubleshooting compliance | Proactive corrective actions and internal audits reduce repeat compliance failures by 70%. |
Understanding key laboratory standards
Biotech and pharmaceutical research facilities operate under strict regulatory frameworks that ensure data integrity and safety. ISO/IEC 17025 is the global standard for testing and calibration laboratories, establishing comprehensive requirements for quality management and technical competence. This standard covers everything from personnel qualifications to equipment calibration, creating a framework that regulatory bodies worldwide recognize and accept.
Good Laboratory Practice (GLP) focuses specifically on pharmaceutical research environments, emphasizing protocols for conducting safety studies. GLP ensures that studies are properly planned, performed, monitored, recorded, and reported. While ISO/IEC 17025 addresses broader laboratory competence, GLP concentrates on non-clinical safety testing integrity.
Key compliance requirements under ISO/IEC 17025 include:
-
Documented quality management systems with clear policies and procedures
-
Personnel competency assessments and ongoing training programs
-
Validated analytical methods with documented measurement uncertainty
-
Equipment calibration using traceable standards
-
Controlled document management and record keeping
GLP requirements emphasize:
-
Study plans and protocols approved before execution
-
Raw data integrity with secure archival systems
-
Quality assurance unit independent from study conduct
-
Facility inspections and process audits
Both standards share common ground in requiring robust documentation, trained personnel, and validated processes. Understanding these overlaps helps labs streamline compliance efforts rather than treating them as separate initiatives. These frameworks protect research integrity, ensure regulatory acceptance of results, and support successful product development pathways.
Prerequisites: What you need before starting
Successful compliance implementation requires foundational elements in place before you begin formal audit preparation. Missing these prerequisites accounts for most audit failures and creates unnecessary stress during inspections.
Complete, current documentation forms the backbone of compliance. Standard Operating Procedures (SOPs) must exist for every critical laboratory process, from sample handling to equipment maintenance. These documents need version control, approval signatures, and accessibility to all relevant staff. Laboratory compliance resources provide templates and guidance for developing comprehensive documentation systems.
Staff competency represents another critical prerequisite. Every team member must understand their roles within the quality system. Document competency through:
-
Initial training records with signed acknowledgments
-
Practical assessments demonstrating technique proficiency
-
Annual refresher training on updated procedures
-
Role-specific qualification matrices
Equipment calibration protocols establish measurement reliability. Every instrument affecting data quality requires documented calibration schedules using traceable standards. Preventive maintenance logs demonstrate proper care and identify potential issues before they compromise results.

Pro Tip: Create a compliance checklist mapping each standard requirement to your current processes. This gap analysis reveals exactly what needs attention and prevents overlooking critical elements during implementation.
Establishing audit trail systems early saves significant time later. Digital systems that automatically timestamp entries, prevent retroactive changes, and maintain version histories provide robust evidence of compliance. Manual systems require additional controls like ink color specifications and countersignature requirements that increase administrative burden.
Missing these prerequisites leads to rushed preparations, incomplete evidence during audits, and findings that could have been prevented. Invest time building these foundations properly, and subsequent compliance maintenance becomes manageable rather than overwhelming.
Step-by-step quality control implementation
Implementing effective quality control aligned with laboratory standards requires systematic execution of specific steps. This structured approach ensures nothing falls through the cracks.
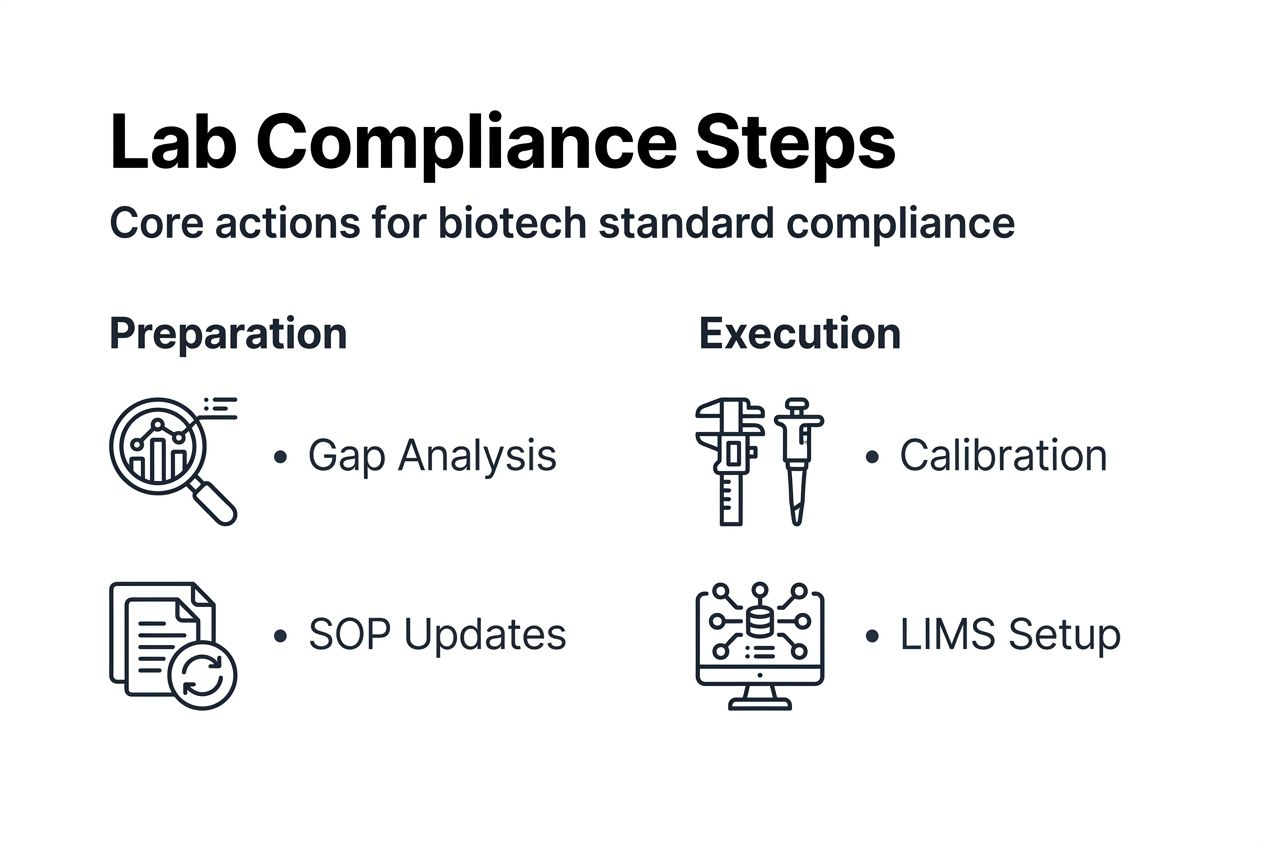
Step 1: Conduct a comprehensive gap analysis benchmarking current processes against ISO/IEC 17025 and GLP requirements. Review every procedure, document, and practice area. Identify specific deficiencies and prioritize them based on regulatory impact and resource availability. This analysis creates your implementation roadmap.
Step 2: Update Standard Operating Procedures and train personnel accordingly. Revise documents to address identified gaps, ensuring clarity and practical applicability. Schedule training sessions covering both technical procedures and quality system understanding. Test comprehension through practical demonstrations and written assessments.
Step 3: Implement validated analytical techniques for peptide quality verification. High-Performance Liquid Chromatography (HPLC) and mass spectrometry provide reliable purity and identity confirmation. Validate these methods by demonstrating accuracy, precision, linearity, and specificity. Document validation parameters and establish acceptance criteria.
Step 4: Deploy a Laboratory Information Management System (LIMS) to enhance data management. LIMS reduces transcription errors, maintains audit trails automatically, and improves data retrieval speed. Consider laboratory quality control solutions that integrate seamlessly with existing workflows.
| Documentation Method | Benefits | Limitations |
| Manual Paper Records | Low initial cost, familiar to staff | High error rate, difficult retrieval, poor audit trails |
| Spreadsheet Systems | Flexible, moderate organization | Version control issues, manual entry errors, limited automation |
| LIMS Implementation | Automated audit trails, reduced errors, fast retrieval | Higher initial investment, training requirements |
Step 5: Establish routine calibration and preventive maintenance schedules. Create calendars tracking due dates for every instrument. Use external calibration services for primary standards and maintain internal secondary standards for routine checks. Document every calibration event with certificates and any corrective actions taken.
Verify peptide products through the entire synthesis and purification workflow. Access peptide products catalog specifications to understand quality expectations and analytical requirements.
Pro Tip: Schedule equipment calibrations quarterly rather than annually. This frequency catches drift earlier, prevents data invalidation, and demonstrates robust quality commitment during audits. Most audit findings related to equipment stem from missed calibrations or expired certificates.
Monitor quality control trends monthly. Plot control chart data identifying shifts or trends before they cause out-of-specification results. Address systematic issues proactively rather than reacting to failures.
Documentation and record-keeping best practices
Documentation proves compliance exists and processes function as intended. Without clear, accurate records, even excellent laboratory practices cannot demonstrate regulatory adherence.
Certificates of Analysis (CoAs) serve as primary evidence confirming peptide purity and compliance with specifications. Each CoA must include batch identification, testing date, analytical methods used, results obtained, and acceptance criteria. Link results to specific equipment and analyst identifications for complete traceability.
Batch record keeping ensures every synthesis step is documented contemporaneously. Records should capture:
-
Starting materials with lot numbers and receipt dates
-
Synthesis conditions including temperatures, times, and reagent volumes
-
Purification parameters and yield calculations
-
Analytical testing results with instrument identification
-
Deviations and corrective actions taken
Organized, accessible documentation supports regulatory inspections effectively. Store records logically with clear indexing systems. Electronic systems should include search functionality and controlled access based on role. Paper systems require fireproof storage and duplicate copies for business continuity.
Laboratory documentation standards provide templates and examples meeting regulatory expectations. These resources help establish consistent formatting and completeness across all record types.
Common documentation pitfalls include:
-
Incomplete entries missing critical data points
-
Retroactive additions without proper justification
-
Unclear handwriting in manual systems
-
Missing signatures or dates on approvals
-
Poor version control on updated procedures
Solutions involve implementing document checklists ensuring completeness, establishing electronic systems with forced fields, and conducting regular documentation audits. Assign document owners responsible for maintaining specific record types, creating accountability.
Integrating LIMS improves accuracy dramatically by eliminating manual transcription and enforcing data entry standards. Automated timestamping and user tracking provide robust audit trails without additional effort. Retrieval speed during inspections reduces stress and demonstrates organizational competence.
Regular documentation reviews identify systemic issues before audits. Monthly spot checks of random records reveal training needs or procedural gaps requiring attention.
Common mistakes and troubleshooting
Understanding frequent compliance failures helps labs avoid repeating mistakes that delay accreditation and waste resources. Learning from these pitfalls accelerates successful implementation.
Top compliance failure points include:
-
Incomplete documentation missing critical traceability elements
-
Insufficient training leaving staff unprepared for quality system responsibilities
-
Missed equipment calibrations invalidating data collected during gaps
-
Ignoring corrective actions from previous audits showing lack of continuous improvement
-
Manual record-keeping errors from transcription mistakes or illegible entries
Corrective action plans (CAPs) mitigate repeated failures when properly implemented. Each CAP must identify root cause, not just symptoms. Address systemic issues through procedural changes, additional training, or process redesign. Document effectiveness checks verifying corrections actually solved problems.
Ongoing staff retraining reinforces quality culture and addresses knowledge gaps revealed through audits. Schedule refresher sessions quarterly rather than only during onboarding. Rotate training topics covering different standard aspects each quarter, building comprehensive understanding over time.
Internal audits serve as preventive measures identifying issues before external assessments. Conduct mock audits using external consultants or trained internal auditors from different departments. Treat findings seriously and implement corrections promptly.
Pro Tip: Maintain a lessons learned log documenting every audit finding, investigation outcome, and corrective action implemented. Review this log during procedure updates and training development. This institutional knowledge prevents repeating mistakes and builds continuous improvement culture.
Laboratories implementing systematic corrective action programs reduce repeat audit findings by 70%, transforming compliance from reactive crisis management to proactive quality assurance.
Leveraging LIMS minimizes manual errors through automated data capture, validation rules, and forced field completeness. Systems alert users to missing information before saving records, preventing incomplete documentation. Consider compliance troubleshooting tips for specific guidance addressing common technical challenges.
Establish cross-functional compliance committees meeting monthly. Include representatives from analytical, quality assurance, and operations. This team reviews metrics, discusses challenges, and coordinates improvement initiatives. Shared ownership of compliance outcomes engages entire organizations rather than isolating responsibility within quality departments.
Safety and regulatory considerations
Safety protocols and regulatory requirements form essential components of holistic laboratory compliance. Separating these elements from quality systems creates gaps that auditors identify immediately.
Chemical and biohazard safety practices protect personnel while ensuring sample integrity. Implement:
-
Personal protective equipment requirements documented in SOPs
-
Chemical inventory management with Safety Data Sheets accessible
-
Spill response procedures with materials readily available
-
Biological safety cabinets validated and certified annually
-
Waste disposal protocols meeting environmental regulations
Temperature-controlled storage maintains sample stability and meets regulatory requirements. Peptides typically require storage between negative 20 to negative 80 degrees Celsius. Monitor storage units continuously with alarmed systems alerting staff to deviations. Document temperature logs daily and investigate any excursions immediately.
Regulatory safety compliance directly impacts audit outcomes and organizational liability. OSHA inspections, EPA compliance, and biosafety protocols receive scrutiny during accreditation assessments. Deficiencies in safety areas raise questions about overall laboratory competence.
Facilities adopting comprehensive safety protocols report zero safety incidents and 40% fewer audit findings in related areas, demonstrating that safety excellence correlates with overall quality system maturity.
Common safety violations include:
-
Inadequate chemical labeling and storage segregation
-
Missing or expired emergency equipment like eyewash stations
-
Incomplete hazard assessments for new procedures
-
Insufficient personal protective equipment for specific hazards
-
Lack of documented safety training for all personnel
Prevention strategies involve regular safety audits separate from quality audits, engaging safety committees, and fostering reporting cultures where near-misses are investigated without blame. Transparency about safety issues demonstrates organizational commitment to continuous improvement.
Vendor registration standards impact supply chain compliance. Verify suppliers meet quality standards through audits or certifications. Maintain approved vendor lists with qualification documentation. Laboratory safety protocols outline requirements for secure supply chains supporting compliant operations.
Integrate safety metrics into quality dashboards. Track incident rates, training completion, and equipment certification status alongside quality indicators. This holistic view ensures balanced attention across all compliance dimensions.
Alternative compliance approaches and tradeoffs
Laboratories can choose different compliance pathways depending on their operational context, regulatory requirements, and resource availability. Understanding options helps select optimal approaches.
Standard compliance follows established frameworks like ISO/IEC 17025 or GLP precisely as written. This approach offers clear expectations, established audit criteria, and widespread regulatory acceptance. Implementation requires significant documentation, formal procedures, and structured quality systems. The rigidity provides consistency but may feel bureaucratic for smaller operations.
Risk-based quality management adapts compliance intensity to actual risk levels. High-risk processes receive more controls, documentation, and oversight than low-risk activities. This approach offers flexibility, directing resources where they matter most. However, it requires sophisticated risk assessment capabilities and may face regulatory scrutiny regarding decisions to reduce controls.
| Approach | Characteristics | Pros | Cons |
| Standard Compliance | Full implementation of ISO 17025 or GLP requirements | Clear expectations, broad acceptance, established audit criteria | High documentation burden, less flexibility, resource intensive |
| Risk-Based Management | Controls proportional to risk levels | Resource efficiency, flexibility, focused efforts | Requires risk assessment expertise, potential regulatory questions, documentation complexity |
| Hybrid Model | Core compliance with risk-based enhancements | Balance of structure and flexibility | Requires careful design, may confuse staff without clear boundaries |
Negotiating regulatory acceptance of alternative compliance models presents challenges. Regulators trained in traditional frameworks may question deviations. Building acceptance requires:
-
Demonstrating equivalent control through alternative means
-
Providing robust justification for each deviation
-
Maintaining comprehensive documentation of risk assessments
-
Engaging regulators early in approach design
Lab maturity influences optimal compliance strategy selection:
-
Startup labs benefit from risk-based approaches, building systems as operations scale
-
Established facilities transitioning to accreditation should adopt standard compliance for clearer pathways
-
Mature organizations with existing quality systems can optimize through risk-based refinements
Consider these criteria when selecting compliance strategies:
-
Regulatory environment strictness and flexibility
-
Available resources for documentation and training
-
Organizational quality culture maturity
-
Product and process complexity levels
-
Customer and stakeholder expectations
Hybrid models combining standard compliance for critical areas with risk-based approaches for supporting processes often provide practical balance. This strategy maintains regulatory confidence while improving efficiency. Explore advanced compliance strategies for detailed implementation guidance.
Review compliance approach effectiveness annually. As organizations mature and processes stabilize, strategies should evolve. What works during rapid growth may need refinement once operations stabilize.
Expected results and outcomes
Understanding realistic timelines and success metrics helps set appropriate expectations and maintain momentum during compliance implementation.
ISO/IEC 17025 accreditation typically requires 6 to 12 months from initial gap analysis to certification. This timeline assumes adequate resources, management commitment, and absence of major process redesigns. Factors extending timelines include:
-
Complex testing portfolios requiring extensive method validation
-
Significant documentation gaps necessitating comprehensive SOP development
-
Staff turnover during implementation disrupting continuity
-
Limited internal expertise requiring external consultant engagement
Success metrics demonstrating effective compliance implementation include:
-
Documentation error rates decreasing by 60% or more
-
Audit findings reducing from double digits to fewer than 5 per assessment
-
Equipment calibration compliance reaching 100% on-time completion
-
Staff training completion rates exceeding 95% within scheduled timeframes
-
Peptide purity consistency improving with tighter specification ranges
Positive impacts on research integrity manifest through enhanced data reliability, improved reproducibility, and stronger regulatory confidence. Publications based on compliant laboratory data receive less scrutiny. Collaborative partnerships strengthen as partners recognize quality system rigor.
Regulatory standing improves measurably. Facilities with accreditation face fewer inspection findings and shorter inspection durations. Regulatory agencies trust data more readily, accelerating approval processes for research applications.
Corrective action effectiveness reduces repeat findings dramatically. Organizations implementing systematic root cause analysis and verification processes see 70% reductions in recurring issues. This improvement demonstrates mature quality cultures focused on continuous enhancement.
Sustaining compliance requires continuous monitoring and regular audit cycles. Plan for:
-
Internal audits quarterly covering different quality system elements
-
Management reviews semi-annually assessing metrics and improvement opportunities
-
Surveillance audits annually by accreditation bodies
-
Full reassessments every 2 to 3 years depending on accreditation scope
Budget ongoing compliance costs including calibration services, training programs, audit fees, and quality personnel time. Treating compliance as continuous operational investment rather than one-time project ensures long-term success. Organizations viewing compliance as embedded in operations rather than separate overhead demonstrate superior sustainability and fewer audit surprises.
Enhance your lab compliance with Bionova Peptides solutions
Maintaining laboratory compliance requires reliable suppliers providing consistent, high-quality materials with robust documentation supporting your quality systems.
Bionova Peptides delivers laboratory reports and certificates verifying purity, identity, and quality for every batch. These Certificates of Analysis provide the traceability documentation auditors expect, supporting your compliance verification processes. Each product undergoes rigorous HPLC and mass spectrometry analysis, with results exceeding 99% purity standards.
Explore comprehensive peptide products synthesized using advanced techniques and validated analytical methods. Temperature-controlled shipping ensures sample integrity from our facility to your laboratory. Visit the Bionova Peptides home page to discover how our quality assurance processes align with your compliance requirements, providing reliable research materials that support your regulatory success.
Frequently asked questions
What are the key laboratory standards biotech labs must comply with?
ISO/IEC 17025 and Good Laboratory Practice (GLP) represent the primary standards governing biotech laboratory operations. ISO/IEC 17025 establishes requirements for testing and calibration competence, covering quality management, technical operations, and personnel qualifications. GLP focuses specifically on non-clinical safety studies, emphasizing protocol adherence and data integrity. Both require documented procedures, trained personnel, validated methods, and calibrated equipment. Meeting these standards ensures regulatory acceptance of research data and supports product development pathways.
How can I prepare my lab staff for compliance audits effectively?
Conduct regular training sessions covering applicable standards, Standard Operating Procedures, and quality system responsibilities. Assess competency formally using written tests and practical demonstrations, documenting results in personnel files. Create role-specific training matrices identifying required competencies for each position. Schedule quarterly refresher training rotating through different compliance topics. Encourage a culture where quality is everyone’s responsibility, not just the quality department’s concern.
What are the common pitfalls causing compliance audits to fail?
Incomplete or missing documentation accounts for the majority of audit findings, including gaps in batch records, missing calibration certificates, or unsigned procedures. Inadequate staff training leaves personnel unable to explain processes or quality system roles during auditor interviews. Neglected equipment calibration invalidates data collected during lapsed periods. Ignoring previous corrective actions demonstrates lack of continuous improvement commitment. Manual record-keeping errors from transcription mistakes or illegible handwriting create traceability gaps that auditors cannot accept.
What role does documentation play in laboratory compliance?
Documentation provides the objective evidence proving your laboratory operates according to established procedures and meets standard requirements. It includes Certificates of Analysis confirming product quality, batch records demonstrating process control, and Standard Operating Procedures defining consistent practices. Complete, accurate documentation supports audit readiness by answering auditor questions through records rather than relying solely on verbal explanations. Organized documentation systems enable quick retrieval during inspections, demonstrating operational competence and reducing audit stress.
